Cell line quality plays a major role in determining the cost of cultivated meat. High-quality cell lines improve efficiency across production, reducing waste, media consumption, and infrastructure needs, which directly impacts the final price consumers pay. Key factors like genetic stability, proliferation efficiency, and differentiation capability influence production costs significantly.
- Genetic Stability: Stable cell lines minimise waste and reduce costly interventions.
- Proliferation Efficiency: Faster-growing cells lower operational expenses by maximising bioreactor productivity.
- Differentiation Capability: Efficient transformation into muscle and fat tissues ensures better use of resources.
For example, optimised cell lines can cut growth media costs by up to 50%, dropping from £120/kg to £70/kg. Companies like Upside Foods have reduced costs by 40% through improved cell line efficiency. By 2030, high-quality cell lines could bring production costs down to £5–£10 per kg, making cultivated meat competitive with conventional options like premium beef (£15–£25 per kg).
Standardising cell lines remains challenging due to variability and technical hurdles, but advancements like immortalised lines and continuous manufacturing are paving the way for lower costs and broader accessibility.
ICAN Webinar about Cell Lines and Culture Media for Cultivated Meat Applications
sbb-itb-c323ed3
Key Cell Line Quality Factors That Affect Production Costs
When it comes to commercial cultivated meat production, three key attributes of cell lines play a major role in determining costs: genetic stability, proliferation efficiency, and differentiation capability. Each of these factors impacts a specific part of the production process, and any weakness can cause costs to escalate. Let’s take a closer look at how each factor influences expenses.
Genetic Stability
Genetic stability measures how consistently a cell line retains its characteristics over multiple growth cycles. A stable cell line ensures that the cells don’t unexpectedly transform into unintended tissue types, a problem known as spontaneous differentiation, which wastes both time and resources [1].
The type of cells used makes a big difference here. Pluripotent stem cells, such as embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs), have the advantage of rapid division and the potential to develop into any tissue type. However, maintaining their stability is a challenge. They require careful management and costly media formulations to prevent deviations. On the other hand, adult stem cells are naturally more stable within their specific tissue lineages, but they come with limitations, particularly slower growth rates. For example, it wasn’t until 2018 that researchers successfully developed stable ESC lines for bovine species, highlighting the technical complexity involved [1]. This lack of stability increases costs due to higher waste and the need for more intensive oversight.
Proliferation Efficiency
The speed at which cells multiply, or proliferation efficiency, directly affects production costs. Faster-growing cell lines are more desirable because they allow producers to generate a larger biomass in less time. This maximises the productivity of expensive bioreactors and reduces per-batch operational costs [1].
However, there’s a trade-off. While pluripotent cells divide quickly, they are harder to control. In contrast, adult stem cells like satellite cells - naturally suited for muscle formation - grow more slowly and have limited replication capacity. For instance, there are only 550 satellite cells per 1 mg of mouse muscle tissue [1], making it difficult to scale production from a small sample. To solve this, companies like Dragon Bio and the Roslin Institute are working on immortalised cell lines. These lines combine the stability of adult cells with the extended growth capabilities of pluripotent ones, achieving 60 to over 100 passages [1].
Differentiation Capability
Once cells have multiplied, they need to undergo differentiation - transforming from a proliferative state into mature tissue types like muscle fibres, fat cells, and connective tissues. This step is critical for giving cultivated meat its texture, marbling, and nutritional value. Efficient differentiation ensures that resources are used effectively, while poor differentiation can lead to massive waste. Growth factors alone sometimes account for over 90% of production costs [4].
In January 2024, researchers at Tufts University made strides in addressing this issue. They engineered bovine muscle stem cells to produce their own fibroblast growth factor (FGF), aiming to eliminate the need for costly external FGF. While this modification successfully removed a significant cost driver, the engineered cells grew more slowly and required further refinement to match the differentiation efficiency of unmodified cells [4].
"FGF is not exactly a nutrient. It's more like an instruction for the cells to behave in a certain way." - Andrew Stout, Director of Science, Tufts Cellular Agriculture Commercialisation Lab [4]
Fine-tuning differentiation processes is essential for lowering costs and making cultivated meat production economically viable.
How Cell Line Quality Affects the Price of Cultivated Meat
Cell Line Quality vs. Cultivated Meat Production Costs
The quality of a cell line doesn't just impact what happens inside a bioreactor - it directly influences the final price consumers pay. Every inefficiency at the cellular level ripples through the production chain, increasing costs at every step.
Effect on Growth Media Costs
Growth media is one of the priciest components in cultivated meat production, making up 40–60% of total production costs during early stages [1][2]. Poor-quality cell lines - those with slow growth rates, low viability, or genetic instability - consume significantly more media. Cells that grow slowly or die off quickly require extra nutrients to achieve the same yield.
Inefficient cell lines can drive up media usage by 20–50%, raising costs from around £100–£150 per kg for high-performing lines to £200–£300 per kg for lower-quality ones [1][2]. On the other hand, optimised cell lines with faster doubling times (24–36 hours) and better differentiation capabilities could reduce media costs from £120/kg to £70/kg, as shown by Tufts University’s 2024 cost modelling [3][5]. This difference could determine whether a product is commercially viable or not.
Infrastructure and Monitoring Costs
Underperforming cell lines also lead to higher infrastructure expenses. When cells have poor density or viability, larger bioreactor volumes are needed to meet yield targets. Scaling from a 10,000-litre bioreactor to a 15,000-litre one can cost an additional £1–£5 million per unit [4][6]. On top of that, batch failures - which occur 10–20% more often with low-quality lines - require backup capacity, adding another £20–£50 per kg to costs [4].
Monitoring costs further add to the burden. Inefficient cell lines with unpredictable growth patterns need advanced real-time sensors to track essential bioreactor metrics like pH, oxygen levels, and metabolites. Installing such equipment can cost £10,000–£50,000 per bioreactor [2][5]. Additional operational costs, like customising media and disposing of waste from failed runs, can contribute an extra £15–£30 per kg [2][5]. For example, Mosa Meat’s 2024 trials showed that switching to high-proliferation lines reduced monitoring interventions by 35%, cutting monitoring costs from 15% to 8% of total production expenses [2][5].
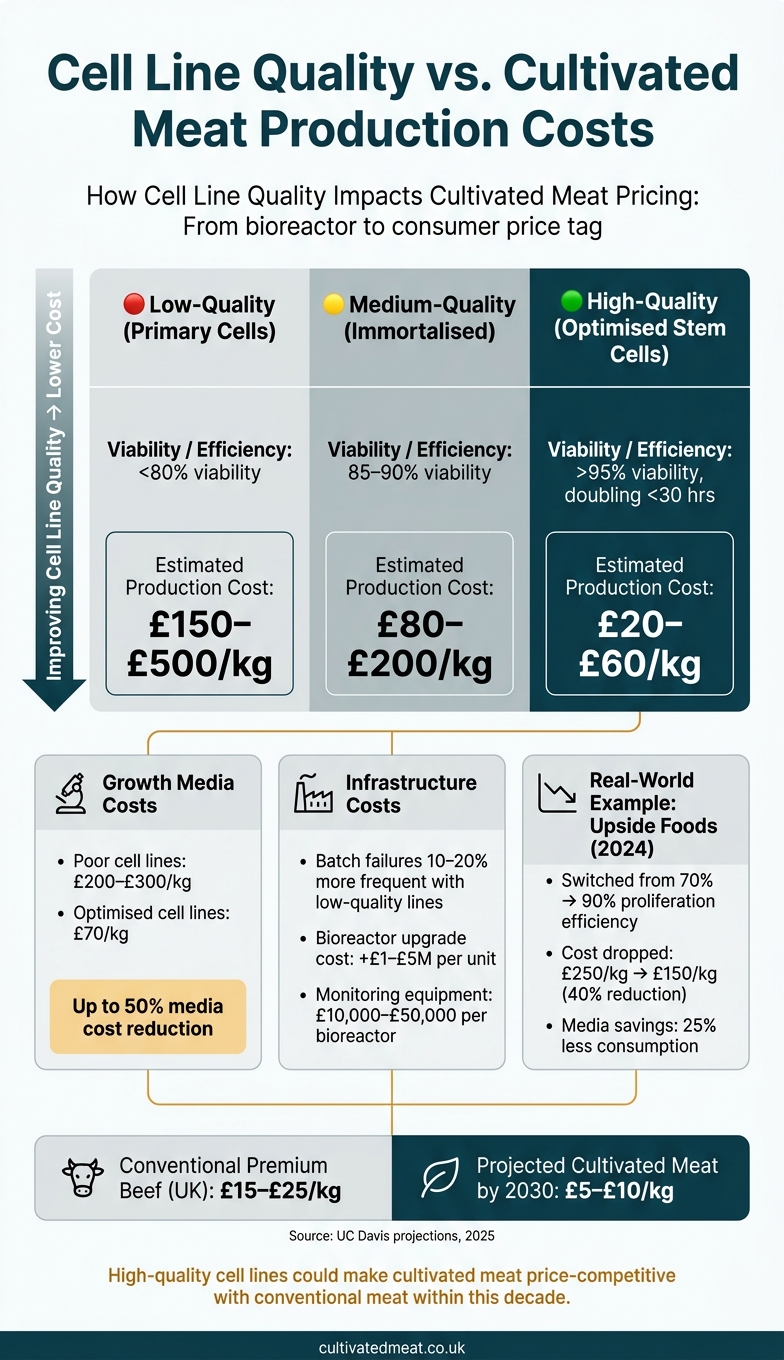
Production Cost Ranges Based on Cell Line Quality
The combined impact of these factors creates a wide range of production costs, depending on the quality of the cell lines used.
| Cell Line Quality Tier | Viability / Efficiency | Estimated Production Cost |
|---|---|---|
| Low-quality (primary cells) | <80% viability | £150–£500/kg |
| Medium-quality (immortalised) | 85–90% viability | £80–£200/kg |
| High-quality (optimised stem cells) | >95% viability, doubling <30 hours | £20–£60/kg |
Looking ahead, projections from UC Davis (2025) suggest that high-quality cell lines could bring costs down to £5–£10 per kg by 2030, thanks to 50–70% savings in media and improved scaling efficiencies [1][3]. For comparison, premium beef in the UK currently sells for around £15–£25 per kg, meaning cultivated meat could realistically match or even undercut traditional meat prices within this decade [1][3].
A real-world example of this trend comes from Upside Foods' 2024 pilot. By switching from avian myoblast lines with 70% proliferation efficiency to genetically stabilised lines at 90% efficiency, they cut production costs by 40%, dropping from £250/kg to £150/kg. These savings were largely due to 25% less media consumption and 15% smaller bioreactor requirements. At commercial scale, costs are projected to drop further to around £40/kg [7].
These examples demonstrate just how critical high-quality cell lines are to making cultivated meat both affordable and scalable.
Barriers to Standardising High-Quality Cell Lines
Even with the clear advantages of high-quality cell lines, achieving standardisation is no small feat. Both technical and logistical challenges make this goal difficult to realise.
Inconsistency in Cell Line Sources
Primary cells are inherently variable. Even samples from the same species can behave unpredictably in a bioreactor, leading to inconsistent yields. This variability makes it tough to establish a production process that is both reliable and repeatable.
The production of cultivated meat adds another layer of complexity. It requires the simultaneous growth and differentiation of multiple cell types - such as myocytes, adipocytes, fibroblasts, and chondrocytes - each with unique quality demands [8]. Ensuring consistency across batches involves stringent quality control at every step of the process.
"A cell line with reproducible and consistent properties is essential for any bioprocess and is crucial for the production of cultivated meat." - Springer Nature [8]
Regulatory requirements further complicate the situation. Transitioning cell lines to animal-component-free (ACF) media while maintaining high-density growth remains a significant technical hurdle, especially at scale [2]. Additionally, risks like cross-contamination and cell line misidentification during this process pose ongoing quality control challenges [8]. These efforts align with broader goals of reducing costs related to growth media and infrastructure.
Approaches to Achieving Standardisation
Several strategies are emerging to address these challenges:
- Spontaneous immortalisation: This method encourages certain cell lines, like chicken and bovine fibroblasts, to proliferate indefinitely without genetic modifications. These lines are stable, high-yield, and well-suited for serum-free production over long periods [2].
- Pluripotent stem cells: Both embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs) offer a consistent starting point and the flexibility to differentiate into various tissue types [8].
- Continuous manufacturing: Using technologies like tangential flow filtration, continuous manufacturing is replacing traditional batch processes. This approach maintains a stable cell environment, reduces variability between batches, and achieves cell densities of up to 130 × 10⁶ cells per ml [2].
In August 2024, researchers from Believer Meats and the Hebrew University of Jerusalem, led by Yaakov Nahmias, demonstrated a continuous manufacturing method for cultivated chicken. They utilised tangential flow filtration with spontaneously immortalised fibroblasts in an ACF medium costing just US$0.63 per litre. This process enabled continuous harvesting for over 20 days [2].
"Continuous manufacturing can offer cost reductions for scaling up cultivated meat production." - Yaakov Nahmias, Director, Grass Center for Bioengineering [2]
These advancements are critical for stabilising production costs and improving efficiency.
What Standardisation Could Mean for Consumer Prices
The potential impact of standardisation on pricing is enormous. Nahmias and his team’s 2024 study modelled a 50,000-litre facility using standardised perfusion technology. Their findings suggested that cultivated chicken could be produced for approximately US$6.2 per pound, a price comparable to organic chicken in retail [2]. Achieving this price point would mark a significant leap towards making cultivated meat a viable option for everyday consumers.
Standardisation also helps cut hidden costs caused by inconsistency, such as failed batches, wasted media, and increased monitoring expenses. By streamlining production, standardised cell lines could directly lower production costs, ultimately benefiting consumers at the checkout.
Conclusion: Lowering Cultivated Meat Costs Through Cell Line Optimisation
The quality of cell lines is a key factor in the cost of producing cultivated meat. Inefficient cell lines drive up expenses by requiring more media and complicating production processes. In contrast, optimised cell lines - those that are stable, efficient, and thrive in animal-component-free (ACF) conditions - can significantly reshape production costs.
A study conducted in August 2024 by Believer Meats and the Hebrew University of Jerusalem highlights this potential. Using spontaneously immortalised fibroblasts, combined with tangential flow filtration and an ACF medium priced at just US$0.63 per litre, researchers estimated a consumer price of around US$6.2 per pound for cultivated meat at scale [2]. This figure places it within the price range of traditional organic meat.
Improvements in advanced cell lines, continuous manufacturing techniques, and affordable ACF media work together to drive down costs. Each advancement amplifies the impact of the others, creating a compounding effect. These efficiencies not only lower production expenses but also reduce resource consumption and environmental impact, making the production process more sustainable overall.
As research progresses and standardisation becomes more feasible, the price gap between cultivated and conventional meat is expected to shrink further. Such developments bring cultivated meat closer to being both affordable and sustainable for everyday consumers. Platforms like Cultivated Meat Shop are keeping a close eye on these breakthroughs, ensuring that consumers stay informed as cultivated meat transitions from a lab innovation to a practical, everyday choice.
FAQs
What defines a high-quality cell line in Cultivated Meat?
A strong cell line in cultivated meat production is one that can self-renew, differentiate into crucial cell types such as myocytes (muscle cells) and adipocytes (fat cells), and consistently retain the traits needed for large-scale manufacturing. These qualities are vital for ensuring dependable and efficient processes, which are the backbone of scaling up cultivated meat production.
Why is growth media so expensive, and how can better cell lines help reduce costs?
Growth media is expensive primarily because of its core ingredients, like recombinant proteins and growth factors. These are typically sourced from costly animal or pharmaceutical origins, often accounting for 55–95% of total production costs. However, advancements in cell lines offer a way to reduce these expenses. By growing more efficiently, using less media, and even producing their own growth factors, these improved cell lines can decrease the need for external nutrients and significantly cut down the overall cost of growth media.
Are immortalised cell lines safe and likely to be approved in the UK?
Immortalised cell lines play a crucial role in producing cultivated meat and are considered safe for use. Techniques like CRISPR and telomerase activation have brought significant progress in genetic engineering, improving the stability and efficiency of these cell lines. Such advancements pave the way for potential regulatory approval in the UK, ensuring a dependable foundation for crafting high-quality cultivated meat products.













