Cultivated meat is still expensive, but costs are dropping fast. Here's how:
- Growth media costs dominate (70%-90%): Fetal bovine serum (FBS) costs £500–£1,000 per litre, making large-scale production impractical.
- Synthetic biology is slashing costs: Engineered microbes now produce growth factors for as little as £1–£5 per litre, cutting media expenses by up to 95%.
- Gene editing boosts efficiency: CRISPR-edited cells grow faster and need less serum, reducing costs by 30%-50%.
- Serum-free media is scaling up: Food-grade alternatives and molecular farming cut costs by 80%-90%.
- Plant-based scaffolds are cheaper: Edible scaffolds from agricultural byproducts lower production costs significantly.
- High-density bioreactors improve yields: These systems increase cell growth efficiency and reduce costs to under £10/kg.
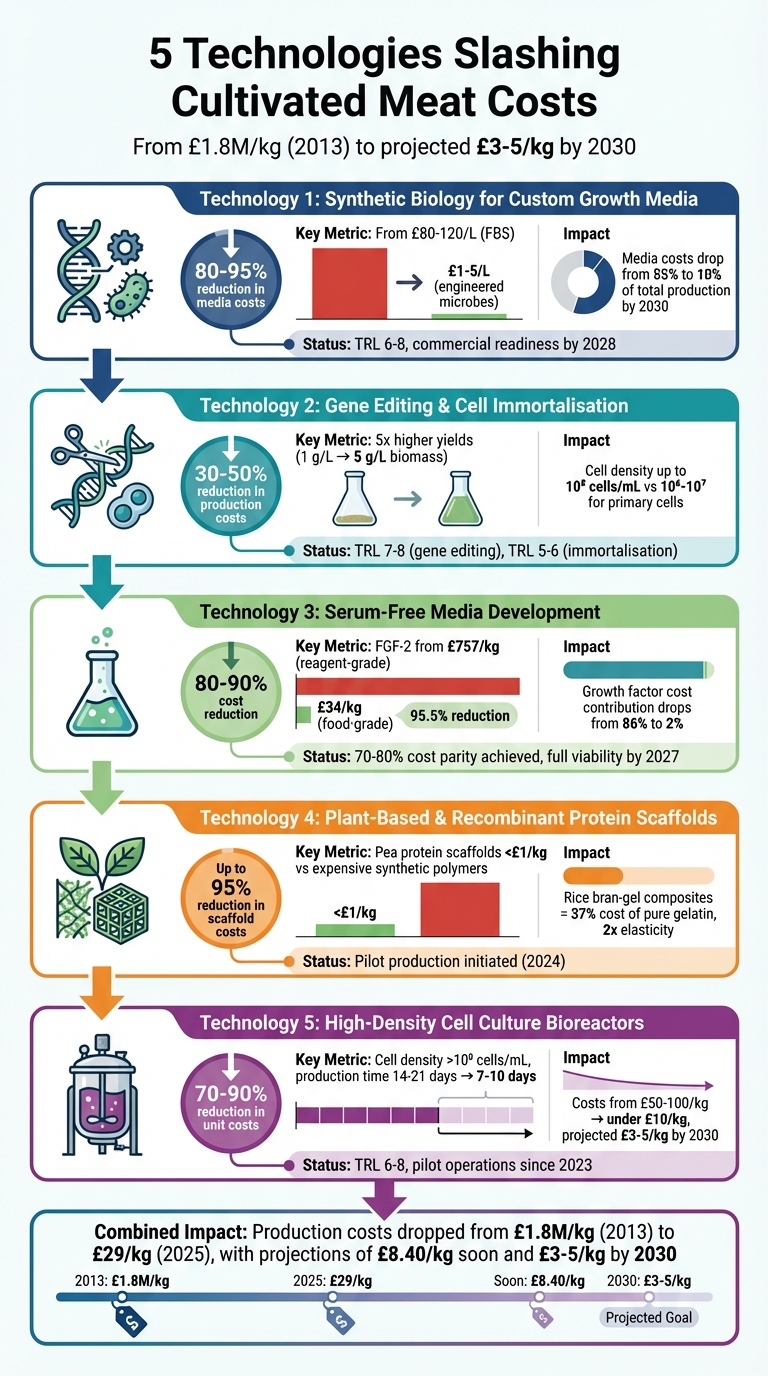
5 Key Technologies Reducing Cultivated Meat Production Costs
1. Synthetic Biology for Custom Growth Media
Cost Reduction Mechanism
One of the biggest challenges in producing Cultivated Meat is the high cost of growth media, but synthetic biology offers a game-changing solution. By engineering microorganisms to produce essential growth factors and nutrients, companies have drastically reduced costs. Traditionally, fetal bovine serum costs between £80 and £120 per litre. Now, with modified microbes, the production cost has dropped to just £1–£5 per litre - an impressive reduction of 80% to 95%. For instance, BioBetter uses plant cell cultures to produce human platelet lysate, cutting costs by 70%. Similarly, Every Company ferments egg proteins to create serum-free media alternatives, partnering with UPSIDE Foods to scale these solutions.
These engineered microbes can synthesise proteins, hormones, and growth factors at a fraction of the cost. Geltor, for example, produces collagen with a 90% cost reduction. Such advancements are not only cutting expenses but also paving the way for more scalable production methods.
Scalability Potential
Thanks to these cost reductions, microbial fermentation has the capacity to scale and meet commercial demand. Unlike traditional methods reliant on animal-derived components, bioreactors enable continuous production of media components, producing tonnes annually. This shift allows the industry to move beyond batch limitations, significantly improving scalability. Vow's ASSet platform exemplifies this progress, reducing media costs from £50–£100 per litre to under £5 per litre at commercial production levels.
Currently, the technology is at TRL (Technology Readiness Level) 6–8, with companies like Nourish Ingredients already piloting molecular farming of growth factors. Full-scale readiness (TRL 9) is projected by 2028, contingent on regulatory approvals for food-grade applications. This ability to manufacture at scale will directly lower production costs, making Cultivated Meat more accessible to consumers.
Impact on Consumer Pricing
These breakthroughs in cost reduction and scalability have a direct impact on consumer prices. Growth media typically account for 40% to 80% of production costs. By slashing media expenses, the price of Cultivated Meat could drop from its current pilot-level range of £50–£100 per kilogramme to an estimated £10–£20 per kilogramme. Projections for 2030 suggest media costs could decrease from 55% to just 10% of total production costs through continued optimisation. Companies like Eat Just are pushing this even further, aiming for price parity with chicken by 2030, targeting a range of £5–£10 per kilogramme.
"Synbio will 'democratise media access, slashing costs 20-fold within five years'", says Dr Ido Hebrew, CTO of Believer Meats.
These advancements not only make Cultivated Meat more affordable but also bring it closer to becoming a viable option for the average consumer.
2. Gene Editing and Cell Immortalisation
Cost Reduction Mechanism
Gene editing, much like synthetic biology, is making strides in reducing production costs for Cultivated Meat, bringing it closer to being affordable for everyday consumers. Tools like CRISPR-Cas9 are playing a pivotal role by enabling the genetic modification of animal cells to replicate indefinitely - a process known as cell immortalisation. This eliminates the need for frequent tissue biopsies, which are both expensive and time-consuming.
Take Upside Foods, for example. By using CRISPR-Cas9 to immortalise bovine satellite cells, they achieved five times higher yields - going from 1 g/L to 5 g/L biomass - in just three months using pilot bioreactors. This innovation cut media costs by 35% and led to the first US regulatory approval for Cultivated Meat chicken. These edited cells were later scaled to 500-litre bioreactors, maintaining an impressive 90% cell viability. A 2023 Tufts University study highlighted that gene-edited bovine cells reduced growth media costs by 40%, largely due to lower serum requirements. Even better, these cells can thrive in serum-free environments, significantly reducing dependence on costly animal-derived inputs. Together with synthetic biology, these genetic advancements are driving production costs down further.
Scalability Potential
Immortalised cell lines offer significant advantages over primary cells, achieving densities of up to 10⁸ cells/mL compared to the 10⁶–10⁷ range for primary cells. Aleph Farms demonstrated this potential by immortalising bovine fibroblasts, which tripled proliferation rates and shortened doubling times from 24 hours to just 8 hours in six-week trials. This resulted in a 25% reduction in production costs and a successful 10 kg pilot batch.
Similarly, CRISPR-edited chicken cells have shown 2.5 times faster growth rates, with doubling times reduced from 48 hours to 19 hours. This progress is crucial for scaling production from lab-sized setups to industrial bioreactors holding tens of thousands of litres. While gene editing has reached a mature stage (TRL 7–8), cell immortalisation is still emerging (TRL 5–6) and faces some hurdles in scaling. However, advancements like these are setting the stage for more affordable Cultivated Meat, as will be discussed in the next section.
Impact on Consumer Pricing
Projections suggest that cell immortalisation could slash Cultivated Meat production costs by 30–50% by 2030. This could bring prices down from the current £10–£20 per kilogramme to under £5. For context, production costs have already dropped dramatically - from £1.8 million per kilogramme to £29 - with further reductions to around £8.40 on the horizon.
Finless Foods has also made notable progress. By applying CRISPR to immortalise bluefin tuna cells, they achieved a fourfold increase in cell density (up to 8×10⁷ cells/mL) and cut costs by 28% during 100-litre production runs. This breakthrough made it possible to host the first tasting of Cultivated Meat seafood. As these technologies continue to improve and gain regulatory approval, the dream of affordable Cultivated Meat is becoming increasingly achievable.
3. Serum-Free Media Development
Cost Reduction Mechanism
Recent strides in synthetic biology and gene editing are driving down the costs of serum-free media (SFM), a critical component in Cultivated Meat production. SFM typically represents at least 50% of the variable operating costs in this industry. For instance, in formulations like Essential 8 medium, a staggering 98% of the total cost comes from just two ingredients: FGF-2 and TGF-β.
To tackle these expenses, companies are adopting several cost-cutting strategies. These include replacing pharmaceutical-grade ingredients with food-grade alternatives, using molecular farming techniques (such as producing recombinant proteins in tobacco plants for as little as £0.75 per gramme), and engineering cells to produce their own growth factors. A notable example is Mosa Meat, which, in collaboration with Nutreco between 2023 and 2024, managed to replace 99.2% of its basal cell feed (by weight) with food-grade components while maintaining similar cell growth to that achieved with pharmaceutical-grade media. Similarly, BioBetter is working on producing insulin, transferrin, and FGF-2 in tobacco plants at the same competitive price of £0.75 per gramme [7]. These innovations not only slash costs but also pave the way for scalable solutions.
Scalability Potential
The scalability of serum-free media has been further proven by companies like Believer Meats, which has developed formulations costing just £0.47 per litre by substituting albumin and fine-tuning nutrient levels. Switching to food-grade basal medium components can lead to significant savings, with overall media costs dropping by around 77%. For example, the cost of L-Arginine plummets from £757 per kilogramme for reagent-grade to a mere £34 per kilogramme for food-grade - a reduction of approximately 95.5% [7].
Impact on Consumer Pricing
These cost-saving innovations are bringing Cultivated Meat closer to price parity with traditional meat products. Companies like GOOD Meat and Aleph Farms have made significant progress in this area. GOOD Meat received regulatory approval from the Singapore Food Agency in January 2023 for its serum-free Cultivated Meat chicken, while Aleph Farms secured similar approval from Israel's Ministry of Health in January 2024 for its serum-free Cultivated Meat beef.
Advances in serum-free media are playing a crucial role in reducing overall production costs. For instance, recombinant growth factors produced through molecular farming could lower their cost contribution in media from 86% to just 2%. These breakthroughs are making affordable Cultivated Meat a more realistic option for consumers [7].
Cost drivers of cultivated meat production
sbb-itb-c323ed3
4. Plant-Based and Recombinant Protein Scaffolds
In addition to advancements in media optimisation and cell engineering, scaffold technologies are playing a crucial role in bringing down the cost of producing Cultivated Meat.
Cost Reduction Mechanism
Scaffolds act as the structural framework that helps cultivated muscle cells grow into meat-like tissue. Traditional synthetic scaffolds, such as polycaprolactone (PCL), are not edible and require expensive removal processes before the meat can be consumed [8][9].
Edible plant-based scaffolds solve this issue by remaining part of the final product, cutting down on extraction costs. Many of these scaffolds are derived from agricultural byproducts, offering an economical and sustainable alternative. For instance, prolamins - proteins found in crops like corn, barley, and rye - are byproducts of the starch and vegetable oil industries, making them a low-cost raw material [9].
"Since the scaffold was edible, no special or additional procedures were needed to extract it from the final product." – Professor Huang Dejian, National University of Singapore [9]
A 2024 study highlighted that rice bran–gel composite scaffolds reduced material costs to just 37% of the price of pure gelatin systems, while nearly doubling elasticity [10]. Similarly, recombinant proteins produced via microbial fermentation offer an ethical and cost-effective alternative to animal-derived materials, while maintaining excellent compatibility with cells [8][10].
Scalability Potential
Plant proteins and polysaccharides, such as alginate and cellulose, are widely available, consistent in quality, and have long shelf lives. These traits make them far better suited for large-scale production compared to specialised medical-grade polymers [8]. In May 2023, researchers at the National University of Singapore, led by Professor Huang Dejian, successfully used prolamins from corn, barley, and rye to 3D-print edible scaffolds. These scaffolds, seeded with pig skeletal muscle stem cells, achieved maximum cell counts within just 11 days, resulting in cultured pork with a texture comparable to traditional meat [9].
"By using readily available cereal prolamins as biomaterials for high-precision 3D printing technology, we open up a new method for manufacturing edible and structured scaffolds to produce cultured muscle meat slices with fibrous qualities." – Professor Huang Dejian, Deputy Head of the NUS Department of Food Science and Technology [9]
Additionally, plant-based scaffolds promote quicker cell growth compared to synthetic polymers. For example, pig cells grown on prolamin scaffolds reached their peak numbers faster than those grown on polycaprolactone [9]. This improved scalability helps further lower production costs, paving the way for more affordable Cultivated Meat.
Impact on Consumer Pricing
The adoption of plant-based and recombinant protein scaffolds has significantly driven down the cost of Cultivated Meat production. Back in 2013, the price stood at an astronomical £1.8 million per kilogramme. By 2025, this figure is expected to drop to around £29 per kilogramme, with predictions suggesting it could fall even further to £8.40 per kilogramme in the near future [1]. This transition - from using non-edible synthetic polymers in the 2010s to edible, plant-derived scaffolds in the 2020s - has not only improved the structural mimicry of meat but has also dramatically reduced production expenses.
5. High-Density Cell Culture Bioreactors
When it comes to cutting down the costs of producing Cultivated Meat, high-density cell culture bioreactors are making waves. These systems are designed to grow a massive number of cells in each batch, which not only boosts output but also reduces the amount of pricey growth media needed - a win-win for efficiency and cost.
Cost Reduction Mechanism
High-density bioreactors are game-changers in terms of cell density, increasing it from around 10⁷ to over 10⁸ cells per millilitre. This leap significantly lowers the consumption of growth media and other reagents. Combined with continuous perfusion techniques, media costs can drop by as much as 50%. For instance, Believer Meats reported a 40% reduction in media costs in their 2024 pilot project, while also doubling their biomass yield [11][12]. Similarly, Mosa Meat, working with bioreactor company CellX, is aiming for 20–30% savings in capital expenditure through advanced high-density systems [3][12].
Scalability Potential
The cost advantages of high-density bioreactors become even more pronounced as they scale up for industrial use. Modular designs make it possible to expand from small lab units of 1–10 litres to massive industrial vessels exceeding 10,000 litres. This can result in a 1,000-fold increase in production capacity [11][3]. Upside Foods has already demonstrated this scalability, producing kilogrammes of cultivated chicken during pilot runs with 2,000-litre bioreactors [11][3].
These systems also dramatically speed up production cycles, cutting them down from the usual 14–21 days to just 7–10 days. At scale, this efficiency can slash unit costs by 70–90%, creating ripple effects that enhance the entire production process [11][2][3].
Impact on Consumer Pricing
Higher yields and scalable production have a direct impact on what consumers pay. By lowering production costs, prices can drop from £50–£100 per kilogramme to under £10 per kilogramme, with forecasts suggesting prices could fall to £3–£5 per kilogramme by 2030 [3][12][5]. Achieving such affordability is crucial for making Cultivated Meat a viable alternative to traditional meat, not just a luxury item.
Technological Maturity
At present, high-density bioreactors are at Technology Readiness Level (TRL) 6–8, meaning they've been successfully tested in commercial settings. Pilot operations have been running since 2023, but full industrial scaling remains at TRL 7 [11][5]. One of the biggest challenges is managing shear stress, which can damage fragile animal cells. Innovations like microbubble oxygenators and 3D scaffold integration are being developed to tackle these issues, ensuring even nutrient distribution and proper oxygen levels [11][4]. These advancements are critical for pushing high-density bioreactors closer to their full potential, making them a cornerstone of cost-effective Cultivated Meat production.
Advantages and Disadvantages
Every technology shaping Cultivated Meat comes with its own set of strengths and challenges, impacting pricing and development timelines.
Synthetic biology has the potential to slash media costs from over 90% to under 10% by 2030. This is thanks to scalable fermentation methods and efforts in commercial-scale recombinant protein production by companies like Geltor [3][4]. However, the high initial costs of research and development, coupled with regulatory complexities for new biologics, have slowed broader adoption. Most applications remain pre-commercial, with only limited pilot-scale data available [3].
Gene editing and cell immortalisation can reduce production times by 50–70% and support high-density cultures. Regulatory approval, such as the FDA’s green light for gene-edited products like Casgevy in 2023, shows promise [2][5]. Still, concerns about off-target edits pose safety risks, requiring thorough validation. Ethical and regulatory hurdles also limit progress, with no Cultivated Meat products using these techniques commercially yet [2][6].
Serum-free media development offers a way to replace costly animal serum - priced up to £100,000 per litre - with defined alternatives. This could cut costs by 80–90%, with companies like Lonza aiming for GMP-compliant trials by 2025 [3][4]. However, these formulations often don’t support cell growth as effectively as serum, leading to lower yields. Customisation for different cell types can also increase costs and extend development time [4].
Plant-based and recombinant protein scaffolds use affordable materials like pea protein (costing less than £1 per kilogramme), potentially reducing scaffold costs by up to 95%. Companies like Ayoba have already initiated pilot production as of 2024 [3]. That said, plant-based scaffolds may not provide the biomechanical properties needed for realistic textures, while recombinant production adds extra biotechnology costs, as the technology is still being refined [4].
Finally, high-density cell culture bioreactors can achieve cell densities exceeding 10⁸ cells per millilitre, boosting yields tenfold and reducing costs to under £5 per kilogramme. BioTech Foods, for example, is targeting 1-kilogramme batch production by 2025 [5]. However, the high capital costs - running into millions of pounds per unit - make this technology less accessible for smaller players. Additionally, challenges like shear stress, oxygen transfer, and contamination risks remain obstacles, even as food-grade adaptations progress [2][6].
Here’s a breakdown of the key points for each technology:
| Technology | Key Advantages | Main Disadvantages | Development Stage |
|---|---|---|---|
| Synthetic Biology for Custom Growth Media | Cuts media costs from >90% to <10% by 2030; scalable fermentation; commercial-scale recombinant proteins (Geltor) [3][4] | High R&D costs; regulatory challenges; limited pilot data [3] | Advanced development; commercial-scale production achieved for some proteins |
| Gene Editing and Cell Immortalisation | Reduces production times by 50–70%; supports high-density cultures; FDA-approved examples (Casgevy, 2023) [2][5] | Safety risks from off-target edits; ethical and regulatory scrutiny; no commercial applications for Cultivated Meat yet [2][6] | Experimental for meat cells; proven in medical fields |
| Serum-Free Media Development | Replaces serum costing up to £100,000/L; 80–90% cost reduction; GMP trials by 2025 (Lonza) [3][4] | Lower performance compared to serum; customisation increases costs; scalability unproven at larger levels [4] | Mid-stage optimisation; GMP trials underway |
| Plant-Based and Recombinant Protein Scaffolds | Affordable materials (<£1/kg for pea protein); 95% cost reduction; pilot production underway (Ayoba, 2024) [3] | May lack texture fidelity; recombinant methods add costs; optimisation ongoing [4] | Early-stage; pilot production initiated |
| High-Density Cell Culture Bioreactors | Achieves >10⁸ cells/mL; tenfold yield boost; costs <£5/kg; 1-kg batches planned (BioTech Foods, 2025) [5] | High capital costs (£millions per unit); technical challenges like shear stress and contamination [2][6] | Late development; proven in pharma, adapting for food production |
These factors not only influence production costs but also determine how quickly Cultivated Meat can become competitively priced for consumers.
Conclusion
The road to affordable Cultivated Meat lies in combining advancements from several key technologies. No single development can drive costs to market-ready levels; instead, the interplay of synthetic biology, serum-free media, and high-density bioreactors holds the most promise for achieving competitive pricing by the decade's end.
Recent pilot studies show that serum-free formulations have already reached 70–80% cost parity, with full commercial viability expected by 2027. At the same time, progress in high-density cell culture bioreactors - scaling production from lab environments to industrial operations - points to significant cost reductions by 2027–2028 [3][4]. For example, the UK-based company Multus Biotechnology is aiming to lower media costs to just £1–£2 per litre by 2028, highlighting the tangible progress being made [4]. However, these advancements must work in harmony to achieve true market parity.
"Continued technical development is essential for achieving £6–£8 per kilogramme parity by the late 2020s." - Dr. Isha Datar, New Harvest
Challenges remain substantial. Regulatory approvals, scaling supply chains, optimising yields, and securing consistent investment - estimated at over £500 million annually - are all critical steps for transitioning from laboratory breakthroughs to large-scale production [3][4]. Yet, with 70% of startups now focusing on synthetic biology as a cost-cutting strategy, the industry is building momentum. By 2030, these efforts could make Cultivated Meat a realistic option for UK consumers [4].
The technologies are evolving at a promising pace, but achieving price parity with traditional meat will demand ongoing collaboration, innovation, and determination.
FAQs
Why is growth media such a big cost driver in Cultivated Meat?
Growth media play a major role in the cost of producing cultivated meat, with growth factors being the most expensive component. These can account for as much as 95% of the total media costs. For example, FGF2, a commonly used growth factor, is priced at approximately £147 per milligram. Such steep costs make large-scale production a financial challenge, underscoring the importance of reducing these expenses to make cultivated meat more affordable.
When will Cultivated Meat reach price parity in the UK?
Cultivated meat is expected to reach price parity in the UK within the next few years. By around 2030, industry advancements aim to make it accessible to the mass market, thanks to continuous efforts in cutting production costs and expanding manufacturing capabilities.
Are gene-edited cell lines safe and likely to be approved for food?
Gene-edited cell lines are deemed safe and are moving towards approval for use in food. Regulatory authorities, including the FDA, evaluate the safety of cultivated meat through thorough pre-market consultations. Recent approvals highlight promising progress, ensuring that cultivated meat adheres to strict safety standards before it becomes available to the public.













